HHS Releases CARES Act Provider Relief Fund Reporting Guidance: Answers Some Questions, Raises More
On Saturday, September 19, the Department of Health & Human Services (HHS) released long-awaited guidance on annual reporting requirements for providers that receive payments from the Provider Relief Fund (PRF). The PRF was established under the Coronavirus Aid, Relief, and Economic Security (CARES) Act and is the distribution vehicle for $175 billion in funding to support hospitals, physicians, skilled nursing facilities (SNFs) and other eligible healthcare providers in response to the COVID-19 pandemic.
This guidance follows a July 20 public notice (as updated August 14 and further amended September 25, now characterized as a “pre-notice”) indicating that individuals or entities that accepted one or more PRF payments exceeding $10,000 in aggregate will be subject to further PRF requirements as a means of complying with PRF terms and conditions. The pre-notice also indicated which PRF distributions would be included in the $10,000 threshold and provided initial indications of timing and deadlines, some of which have since been updated.
The September 19 reporting guidance details the data elements that must be submitted for calendar years (CYs) 2019 and 2020 to report on how the funds were used, defined as:
- Healthcare expenses attributable to COVID-19 that have not been reimbursed by another source and do not need to be reimbursed, including general and administrative (G&A) or healthcare-related operating expenses; and
- PRF payments not fully applied to healthcare expenses attributed to COVID-19 and that were then applied to lost revenues.
The document includes three significant changes to previous guidance:
- Expenses must be net of any other reimbursable sources and PRF payments must be allocated to expenses first, prior to allocation against losses. The guidance appears to suggest that only expenses remaining after all other eligible sources have been exhausted may be considered as reimbursable expenses. The guidance also introduces a required order for allocating funds. PRF payments will first be applied to offset healthcare-related expenses attributable to the coronavirus; if there are PRF payments remaining after allocating them to such expenses, the remainder may be allocated to losses.
- HHS introduces a new definition and methodology to calculate “lost revenue” as a negative change in year-over-year net patient operating income and applies a cap. In an approach that is markedly different from prior guidance, HHS instructs providers to report losses as a change in net patient care operating income from 2019 to 2020 (i.e., patient care revenue less patient care-related expenses). In essence, it changes “lost revenue” to mean “lost profit.” HHS also imposes a cap on losses against a 2019 baseline quarterly operating margin. Recipients that reported negative net operating income from patient care in 2019 may apply PRF amounts to lost revenue up to a net zero gain/loss in 2020.
- Providers that do not expend PRF funds in full by the end of CY 2020 will have an additional six-month period to expend funds in a qualified manner. If a provider has not fully expended its PRF funds in full as of December 31, it may apply remaining amounts toward expenses attributable to the coronavirus (not reimbursed by other sources), or apply those sums toward lost revenues in an amount not to exceed the provider’s 2019 net gains. For example, the reporting period from January to June 2021 will be compared to the same period in 2019. No instruction is provided in the event a provider still has remaining funds after that date, but presumably such funding would need to be returned.
On September 23, HHS also published a summary infographic on reporting requirements.
This guidance provides the greatest level of detail to date on reporting requirements but leaves many questions for providers related both to compliance and overall implications. The reporting process will be managed by the Health Resources and Services Administration (HRSA), and the agency plans to host question-and-answer sessions via webinar and will issue FAQs to aid in the reporting process.
There are sure to be winners and losers under this new rubric, resulting in some providers being required to return potentially significant amounts of funding. It is unclear how this guidance and timeline will relate to future distributions from the PRF. As we await clarifications and further guidance, providers should track their expenses against the expense categories outlined in the guidance, catalog all sources and amounts of coronavirus-related funding from other sources, and begin determining their net operating income changes from 2019 to 2020 on a quarterly basis.
Details
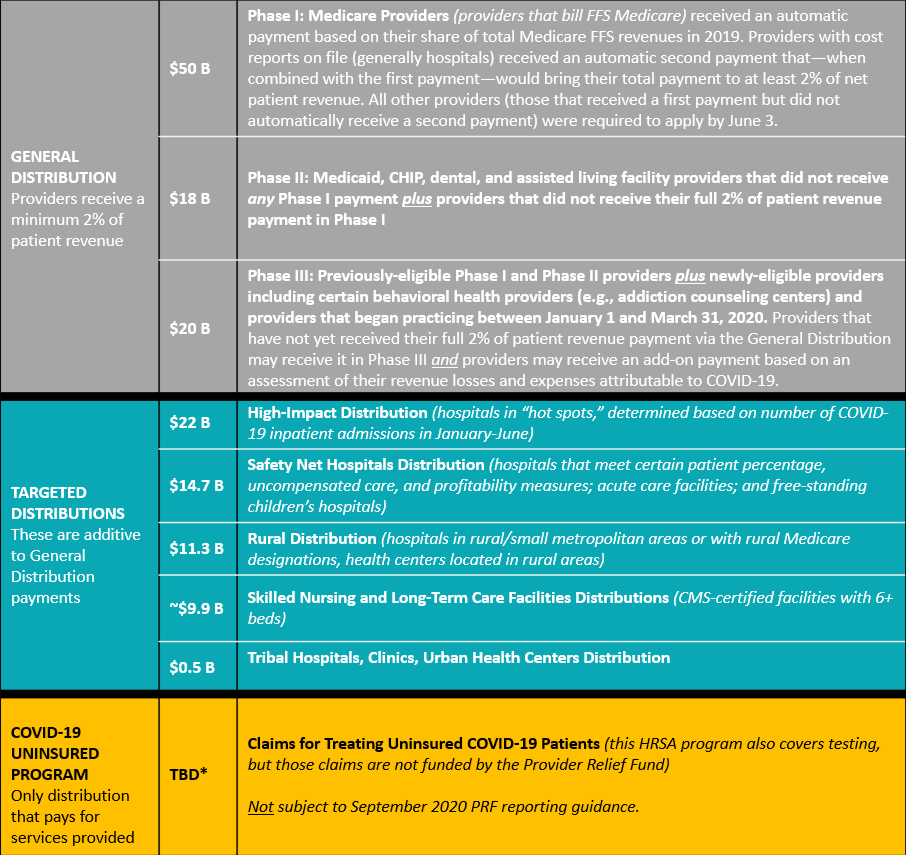
Program Eligibility
Reporting obligations apply to any provider that received over $10,000 in aggregate (total of all payments received) from any of the following PRF distributions (see also reference table at end of this summary):
- General Distributions:
- Phase 1 General Distribution: An initial $30 billion general Medicare Distribution (“round one” – payments that were distributed automatically based on historic Medicare fee-for-service billing) as well as an additional $20 billion general Medicare Distribution (“round two” – supplemental distribution to providers that received a distribution in round one; required an application)
- Phase 2 General Distribution: Medicaid, CHIP, Dental and Assisted Living Distribution (application-based distribution originally open only to providers that were not eligible to receive funds from Medicare in round one or two above but since expanded to allow select Medicare providers to also apply)
- Targeted Distributions:
- High Impact Area Distribution
- Rural Distribution
- Skilled Nursing Facility Distribution
- Indian Health Services Distribution
- Safety Net Hospital Distribution
These particular reporting requirements do not apply to the Nursing Home Infection Control or Rural Health Clinic Testing distributions, nor to reimbursement from the HRSA Uninsured Treatment Program.
Reporting Entities
In general, the entity that receives the PRF payment(s) is the entity required to report, as identified by the Tax Identification Number (TIN) receiving the PRF payment. Reporting entities must submit demographic information including the name of the reporting entity, TIN, fiscal year-end date and federal tax classification. National Provider Identifier (NPI) is listed as an optional category.
In the case of a parent organization with subsidiary TINs, the parent can direct the use of and report on payments distributed to a subsidiary via one of the General Distribution funds (see above). However, if a subsidiary TIN received a Targeted Distribution payment, the subsidiary TIN must report the use of funds for that payment. Further, reporting guidance appears to prohibit the transfer or redirection of funds from a Targeted Distribution payment to any subsidiary legal entities.
Reporting Requirements
In addition to demographic information, entities will also be required to submit information on (1) expenses incurred that were attributable to COVID-19 that were not reimbursed by another source in CY 2020 and (2) information on lost revenues, as defined and detailed below.
Reporting entities will also have to provide specific categories of nonfinancial data, including facility, personnel and patient care metrics (see below).
The financial reporting requirements are bifurcated based on a PRF payment threshold:
- Recipients of Between $10,000 and $499,000 in PRF Payments:
- Healthcare-related expenses attributable to the coronavirus, net of other reimbursed sources (e.g., payments received from insurance and/or patients and amounts received from federal, state or local governments) in two aggregated categories: (1) General and Administrative (G&A) expenses and (2) other healthcare-related expenses.
- Allocation of PRF to lost revenues (see below).
- Recipients of Greater Than $500,000 in PRF Payments:
- Entities will be required to provide more detailed information related to the two expense categories described below, net of other reimbursed sources, according to the subcategories below.
- Allocation of PRF payments to lost revenues.
Calculating Expenses
Expenses attributable to the coronavirus may be incurred in direct patient care overhead activities related to treatment of confirmed or suspected cases of COVID-19 (as defined in the PRF terms and conditions and HHS FAQs), preparing for possible or actual coronavirus cases, and maintaining healthcare delivery capacity, which includes operating and maintaining facilities, etc.
Categories of Healthcare Expenses
| G&A Categories |
Healthcare-Related Expenses Attributable to the Coronavirus |
|
|
Note: The Terms and Conditions associated with each PRF payment do not permit recipients to use PRF money to pay any salary at a rate in excess of Executive Level II, which is currently set at $197,300. For the purposes of the salary limitation, the direct salary is exclusive of fringe benefits and indirect costs. The limitation only applies to the rate of pay charged to PRF payments and other HHS awards. An organization receiving a PRF payment may pay an individual’s salary amount in excess of the salary cap with nonfederal funds.
Allowable expenses include only those not reimbursed or reimbursable by another source. The guidance does not specify what constitutes other sources in the discussion of expenses, though it does list several examples of other sources of funding in its discussion of lost revenues (see below). It also does not comment on the potential for future reimbursements beyond the reporting period.
The two combined categories of expenses minus any such reimbursements or payments from “other sources” will result in the net allowable COVID-19-related expenses for PRF offset.
As noted above, the guidance indicates PRF payments will need to be allocated to these net allowable expenses first, before funding may be applied against losses. As such, it appears to make reporting of COVID-19-related expenses mandatory for providers accepting PRF payments.
New Methodology for Calculating and Reporting Lost Revenues
The CARES Act directs HHS to provide reimbursement to providers for “healthcare related expenses or lost revenues that are attributable to coronavirus” that have not been reimbursed by other sources. While the statute provides some broad guidance on what might constitute eligible expenses, it does not provide guardrails to define lost revenues.
Prior to the September 19 notice, HHS had provided general guidance defining lost revenue via its FAQs, stating that “the term ‘lost revenues that are attributable to coronavirus’ means any revenue that you as a healthcare provider lost due to coronavirus” (FAQ modified 6/19/2020), further suggesting that providers could use “any reasonable method” to calculate lost revenue, including comparing actual revenue in 2020 to actual revenue in 2019, or comparing 2020 budgeted revenue to actual experience.
In a significant departure from this earlier guidance, HHS elects to define lost revenue for purposes of reporting as the difference between 2019 and 2020 “net patient care operating income (i.e., patient care revenue less patient care–related expenses…), net of the healthcare-related expenses attributable to coronavirus.”
Further, HHS imposes a ceiling on the amount of reportable lost revenue under the net operating income methodology. PRF payments may be applied toward lost revenue only up to the amount of the provider’s 2019 net gain from healthcare-related sources. Providers with negative net operating income from patient care in 2019 may apply PRF amounts to lost revenues up to a net zero gain/loss in 2020 (so, if a provider had a negative operating income in 2019, it can retain PRF funding to a breakeven level).
Providers will be required to report revenue/net charges from patient care (prior to netting with expenses) for the CYs 2019 and 2020. CY actual revenues will be entered by quarter (e.g., January–March 2019, April–June 2019, etc.). HHS specifies that “patient care” means healthcare, services and supports as provided in a medical setting, at home or in the community. It should not include (1) insurance, retail or real estate values (except for SNFs, where that is allowable as a patient care cost), or (2) grants or tuition.
Actual revenues/net charges should include patient care revenues from Medicare Parts A, B and C; Medicaid; CHIP; commercial insurance; and self-pay patients, net of uncollectible patient service revenue recognized as bad debts.
Reporting entities will also be required to detail “other assistance received” in CY 2020, including funding from the Treasury, Small Business Administration (SBA) and CARES Act/Paycheck Protection Program (PPP); FEMA CARES Act funding; CARES Act testing; local, state and tribal government assistance; business insurance; and other federal and/or coronavirus-related assistance.
Finally, while PRF funds can be allocated to G&A and healthcare-related expenses associated with the coronavirus incurred only in CY 2020 (as detailed above), for purposes of determining lost revenue, HHS will require a quarterly breakdown of all G&A and healthcare-related expenses for both CY 2019 and CY 2020. A description of the types of elements that can be included in each category is included on page 5 of the notice.
Additional Reporting Requirements
The guidance states providers will additionally be required to provide the following nonfinancial information as collected on a quarterly basis (but the notice does not specify whether this information will be limited to 2020):
- Personnel Metrics: Total personnel by labor category (full time, part time, contract, other: recipient must define), total rehires, total new hires, total personnel separations by labor category.
- Patient Metrics: Total number of patient visits (in person or telehealth), total number of patients admitted, total number of resident patients.
- Facility Metrics: Total available staffed beds for medical/surgical, critical care and other beds.
In the event of a change in ownership, Reporting Entities that acquired or divested of related subsidiaries will need to provide information on the change in ownership, including the date of the transaction, TINs involved in the transaction, percentage of ownership in the entity and whether the reporting provider holds a controlling interest in the entity.
Single Audit Requirements for Certain Recipients
Entities with total aggregated federal expenditures greater than $750,000 in 2020 are subject to Single Audit requirements, as set forth in 45 C.F.R § 75.501.
PRF payments are required to be included in the federal funding that determines whether nonfederal entities meet the $750,000 threshold for Single Audit requirements, in accordance with subsection 75.216 of the Uniform Guidance issued by the Office of Management and Budget (OMB), and whether for-profit entities are required to meet audit requirements, per guidance from HHS released July 22 and modified July 30, 2020.
Providers should be aware that most COVID-19 programs are expected to be subject to Single Audit, so even if a provider receives less than $750,000 from the PRF specifically, its PRF payment will count toward aggregate federal funding for COVID-19. In addition, Single Audit or program audit requirements apply to expended federal awards in excess of $750,000 in a single fiscal year, which includes the total of all federal awards, COVID-19 and non-COVID-19 related. (COVID-19-related funding for providers not subject to Single Audits includes PPP loans and Medicare Accelerated and Advanced Payment Program loans.)
As part of the PRF reporting, providers will need to indicate whether they are subject to Single Audit requirements in 2020 and, if yes, whether the auditors selected PRF payments to be within the scope of the Single Audit (if known at the time the Reporting Entity submits its report).
Key Dates
- December 31, 2020: End of the 2020 CY reporting period.
- January 15, 2021: Online reporting system scheduled to open.
- February 15, 2021: First reporting deadline for all providers on use of funds (45 days after the end of the year). Note, if a provider has used and can appropriately allocate all PRF payments by the end of 2020, it will only have to submit one report.
- June 30, 2021: End of grace period for expenditure of PRF payments.
- July 31, 2021: Second reporting deadline, for expenditure of PRF payments between January 1 and June 30, 2021.
Implications
Potential for Repayment
PRF payments are not a loan requiring repayment after a determined period of time. Neither are they a fixed-amount grant. They are in effect payments to reimburse costs or offset losses and, as such, there has long been some degree of uncertainty about circumstances under which a provider may be required to return some amount of funding. In a FAQ dated 5/6/2020, HHS states, “Generally, HHS does not intend to recoup funds as long as a provider’s lost revenue and increased expenses exceed the amount of Provider Relief funding a provider has received.”
Providers were given 90 days to decide whether to keep the funds and attest to their agreement to adhere to a set of terms and conditions. Most providers did so based on a combination of the initial formulas specific to each distribution and HHS guidance related to calculating lost revenues and expenses attributable to the coronavirus.
However, the decision to impose a net operating income standard rather than a lost revenue calculation may require some providers to return PRF monies, particularly those that may have taken aggressive cost-cutting measures early on to manage the pandemic (such as those that experienced steep revenue drops from the closure of elective services in regions that did not see an equivalent surge in COVID-19 cases). The experience with, timing of and impact of COVID-19 have varied significantly across the country. The new guidance could also be an issue for provider organizations that end their fiscal year in June or September. In a September 25 letter to HHS Secretary Alex Azar, the American Hospital Association outlined scenarios where high-need providers, including rural and safety net providers, could find themselves in a position to return funds that were intended to stabilize and rebuild operations.
The new formula does, however, allow providers that operated in the red in 2019 to break even in 2020, perhaps signaling HHS’ intent that providers not be in a position to profit from relief funds.
Reporting “Grace Period” in 2021
If a provider has accounted for all PRF dollars by the end of 2020, it will submit one report as detailed above. If a provider has remaining funds after December 31, 2020, it will have until the end of June 2021 to use those dollars.
While the guidance imposes a cap on lost revenues, it does not do the same for expenses attributable to the coronavirus. Recipients may spend PRF payments until June 30, 2021.
In the September 24–25 Medicaid and CHIP Payment and Access Commission (MACPAC) meeting, some commissioners expressed concern that this new guidance may send the message that providers should “use it or lose it” by maximizing their expenditures in response to COVID-19 so as not to have to give back any money, rather than using the funds to bolster their financial health and their ability to keep their doors open amid significant revenue losses.
Although not specified, it appears from the guidance that any funds remaining unused at the end of June 2021 will likely need to be returned.
Restrictions on Payment Application Across TINs
The September 19 guidance and some recent PRF FAQs appear to restrict a health system’s options for redirecting or transferring Targeted Distributions between subsidiary legal entities. This may pose challenges for integrated delivery systems and may impose unintended consequences based on a health system’s corporate structure.
While a parent organization can report for subsidiary TINs related to the General Distribution funds, additional guidance is needed as to whether expenses and operating income can be reported in aggregate or whether they will need to be reported at the TIN level.
Separate Quarterly Reporting for Providers Receiving Over $150,000 Not Required
The PRF terms and conditions state that any provider receiving more than $150,000 in coronavirus stimulus funds will need to submit a quarterly report to HHS and the Pandemic Response Accountability Committee. In a FAQ dated June 13, 2020, HHS states that recipients of PRF funding will not need to submit a quarterly report due to public reporting facilitated by HHS and the Department of the Treasury, but that providers will need to submit reports to HHS related to their use of the funding. This guidance appears to fulfill HHS’ requirements for reporting related to use.
Relationship to Future Distributions and PHE Timeline
It is unclear whether the reporting timelines may shift if Congress were to appropriate additional funding for the PRF or if the Public Health Emergency (PHE) period continues to be extended; on October 2 HHS renewed the PHE and it is now in effect until January 20, 2021.
On October 1, HHS announced a new round of General Distribution funding of up to $20 billion, currently called “General Distribution Phase 3” on the HHS PRF website, which will require an application process. The announcement did not indicate whether this new funding opportunity will be subject to the same reporting structure or timeline.
Looking Ahead
While most provider organizations have been keeping close track of their COVID-19-related costs, this reporting guidance provides the most detailed information on categories of eligible expenses to date. Providers should closely review and track what can be included in G&A and healthcare-related expenses.
Providers will also need to begin a detailed quarter-by-quarter review of patient revenues and costs across 2019 and 2020.
As guidance related to the PRF has changed frequently since April and HHS has indicated it plans to develop FAQs specific to reporting, providers should frequently check the PRF “Reporting and Auditing” webpage.
Exhibit: Summary of PRF Distributions Announced to Date